 Noble gases are elements that have a full valence shell, meaning that the outer shell is completely filled with electrons. Thus, it wants to pick up an electron and become a Cl - ion. This can be seen with Chlorine, which in it's neutral state is missing one electron in it's valence electron shell. An atom can also gain an electron (usually to fill it's valence shell) and turn into a negatively charged ion. Thus it wants to lose a single electron and become an Na + ion. For example, sodium (Na) has one electron in it's outer shell. When an electron leaves a neutral atom, it loses a negative charge and turns into a positively charged ion. This can be to create an ionic bond or to become an ion. In chemical reactions, the electrons can even break free from the valence shell. This means that electrons in the inner shells can absorb bits of energy and move (jump) to the valence electron shell. In addition, core electrons in the inner shells have lower energy levels than the valence electrons occupying the outer shell. This difference comes from the electric force being an inverse square law. Valence electrons are the farthest from the positive charge (the protons) and thus tend to be easier to remove than core electrons this means that it takes them less energy to move far away from the atom. Electrons that are closer to the nucleus are in filled orbitals and are called core electrons. Valence electrons are the electrons orbiting the nucleus in the outermost atomic shell of an atom. Each of these orbitals serves to create a shell of electrons in the atom. These orbitals and the energy needed to remove each of these electrons from the atom are set by quantum mechanics. 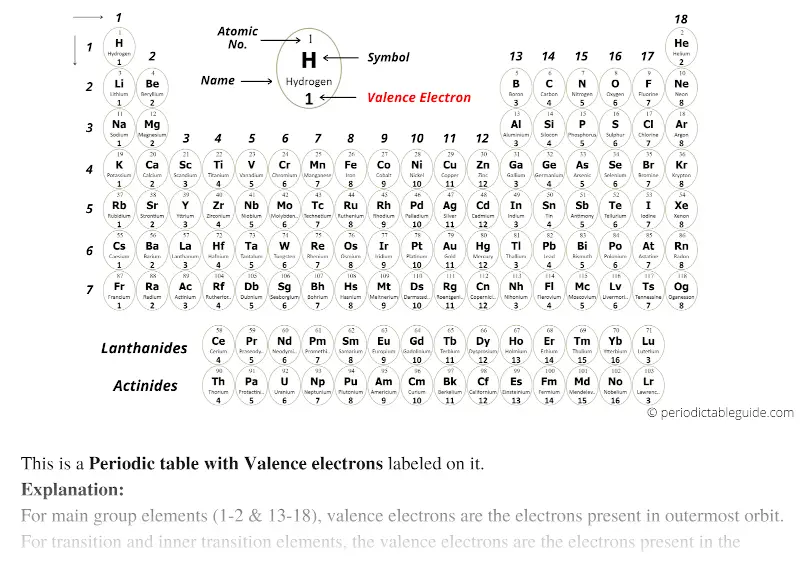
This picture does not address the quantum mechanics of electrons around atoms.Įlectrons exist in orbitals around a nucleus. Figure 1: The two yellow electrons on the outermost oval are the valence electrons the other 10 electrons are core electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
